Safety and tolerability profile1
The safety of SUPPRELIN® LA in children with CPP was evaluated in 2 single-arm clinical trials conducted in a total of 47 patients (44 females and 3 males).
This table shows the implantation adverse reactions of
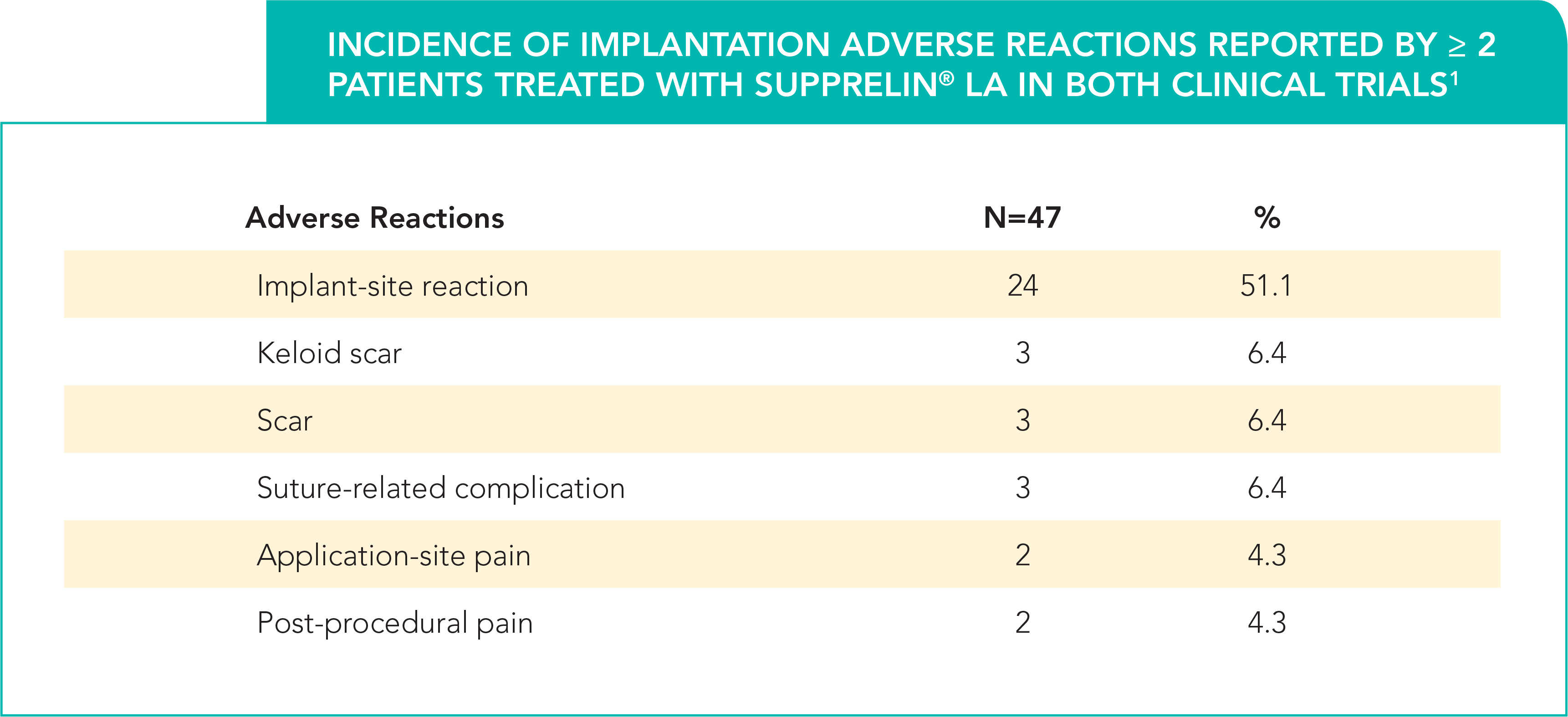
- Implant-site reactions included: discomfort, bruising, soreness, pain, tingling, itching, implant-area protrusion, and swelling1
- One patient discontinued therapy due to infection at the implant site1
- In a Phase 3, 12-month trial, the majority of implant-site reactions were mild to moderate, and most resolved within 1 to 2 weeks without treatment2
- In the long-term extension phase of the trial, the most frequently reported treatment-emergent adverse events reported by 19 patients (52.8%) over 6 years were mild to moderate implant-site reactions3
- In the long-term extension phase, the rate of implant breakage was 22.1% over 6 years3
SERIOUS ADVERSE REACTIONS
Two patients experienced a serious adverse reaction in the 12-month study1,3:
- One patient with Stargardt's disease experienced amblyopia
- One patient had a benign pituitary tumor (pituitary adenoma)
Serious adverse events were reported in the long-term extension phase, including: depression (n=1), aggression (n=2), and apnea and acute bronchopneumonia (n=1). One patient died during the extension phase from acute bronchopneumonia and Dravet syndrome.3
